 
Sulfur is characterized as a non-metal because it is consistent with the 3 physical properties listed for nonmetals. How would you prove that Sulphur is a non-metal? An element that becomes an ion must have free room for its electrons to move and to create an electric charge. It is a poor conductor of heat and electricity, because electrons are not free to move. Sulphur is a non-metal because it is consistent with three physical properties listed for non-metals. It is a poor conductor of heat and electricity because the electrons are not free to move. Because it is consistent with the 3 physical properties listed for non-metals. Because it’s not placed in metal group in the periodic table. Sulphur is a non–metal with symbol S and atomic number 16. Is the element sulfur a metal or nonmetal explain how you got your answer? The non-metallic chemical element sulfur, 3216S, referred to in Genesis as brimstone and identified as element by Lavoisier, is the tenth most abundant element in the universe and the fifth most common element on Earth. Is the element sulfur a metal or non-metal? 5 Why is sulfur a poor conductor of heat and electricity?.4 Is there such a thing as a sulfur metal?.3 Why Sulphur is non-metal give 3 reasons?.2 How would you prove that Sulphur is a non-metal?.1 Is the element sulfur a metal or non-metal?.For example, they are not malleable, they have relatively low density, they are dull, and they are poor conductors of heat and electricity, etc. Non Metals can be defined simply by having the opposite properties of metals. Now that we have established the properties that make most elements on the periodic table into metals, we can now present the complete list of metal elements: Element This is hydrogen that, when exposed to either extremely high or extremely low temperatures, can display some of these common properties. There is one non-metal element that can sometimes act as a metal. Finally, most metal elements lose electrons during reactions.One of the common and, perhaps, most noticeable properties that most metal elements share is that they corrode when exposed to seawater or air.With the exception of potassium, lithium, and sodium, most metals have a high density.Also, most metals can be made into wire.This means that metals are relatively easy to be broken up into sheets. Another important property that many metal elements share is that they are malleable. But have low ionization energies and low electronegativities. Metals also tend to be a good conductor of heat and electricity.Another way to recognize a metal is that they tend to be shiny.Generally, the melting point of metals is high. So it stays at a liquid stay even at room temperature. Mercury is the only metal that is always liquid no matter the temperature. The only exception to this property, in fact, is mercury. Most metals have a solid state when they are at room temperature. “Periodic table blocks spdf (32 column)” by DePiep via Wikimedia Commons is licensed under CC-BY-SA 3.0 What Are The Common Properties Of Metals? Not all properties are shared by all metals, but all elements that shared several of these properties can be considered metals. An element needs to share several of these properties in order to be considered a metal. It is important to become familiarized with these different properties. There are several properties that make most elements metals. We need less memorization – I never memorized the periodic table of the elements – I’ve never used it, and I’m a physicist! I can look it up. There are five different kinds of metals:Īll metal elements are placed together in the periodic table. Before going over the complete list of metals, it is important that we define what a metal is. Knowing which ones are or are not will help us to group them correctly.  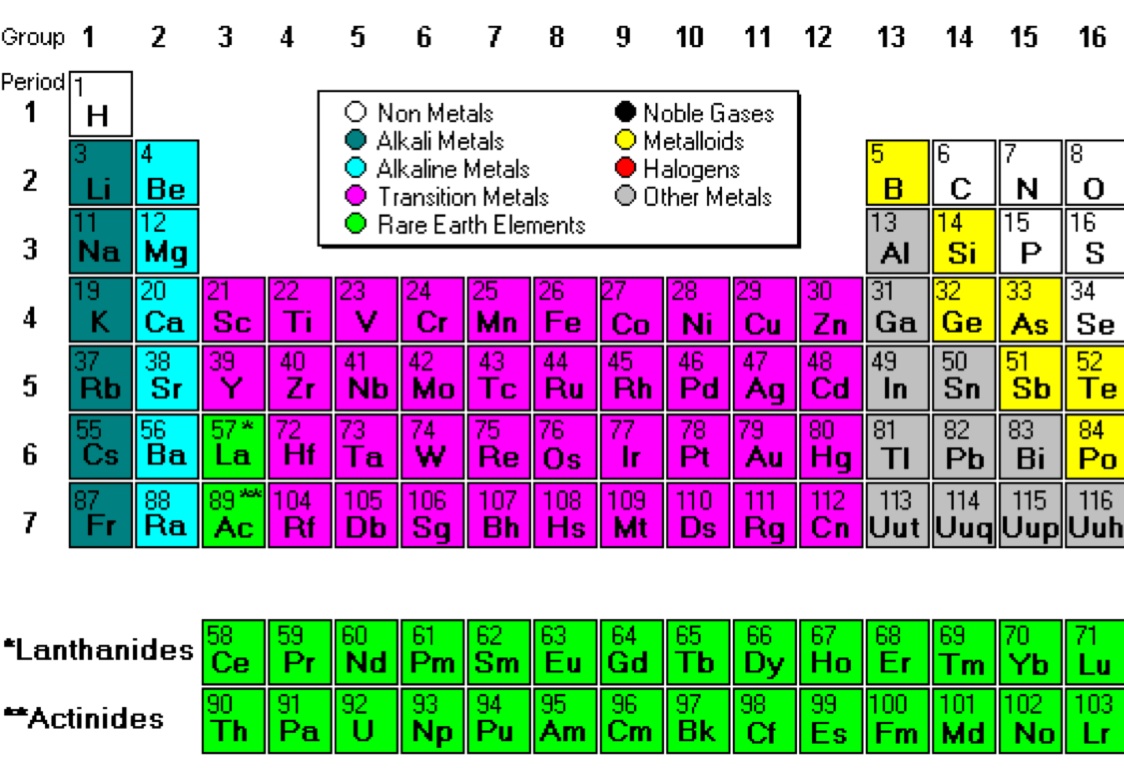
What Is A Metal?Īs stated in the introduction, most elements are metals or, at least, can be considered as such. So, knowing what are those properties is a good way to begin our study. A useful way to approach the study of elements is to distinguish whether they are metal or non-metal. The good news is that most elements are metals. If you are trying to learn to distinguish between metals and non-metals, a list and their uses is a good way to break them down and help memorize the difference between the two. When we study the elements, it is important to know which elements are metals and which ones are not. The nonmetals list which makes up the periodic table includes hydrogen, helium, carbon, sulfur, nitrogen, oxygen, radon, neon, other halogens, and noble gases etc. The metals list which makes up the periodic table includes iron, lead, gold, aluminum, platinum, uranium, zinc, lithium, sodium, tin, silver, etc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
